In the realm of catalysis, the efficiency and effectiveness of reactions are paramount, especially when we consider the need for sustainable energy solutions. Recent advancements in the field, particularly those reported by a research team from the University of Science and Technology of China (USTC), have unveiled significant insights into the behavior of metal loading in single-atom catalysts. The implications of this research extend far beyond academic curiosity; they are pivotal for industries aiming to harness catalysis for energy generation and pollution reduction.
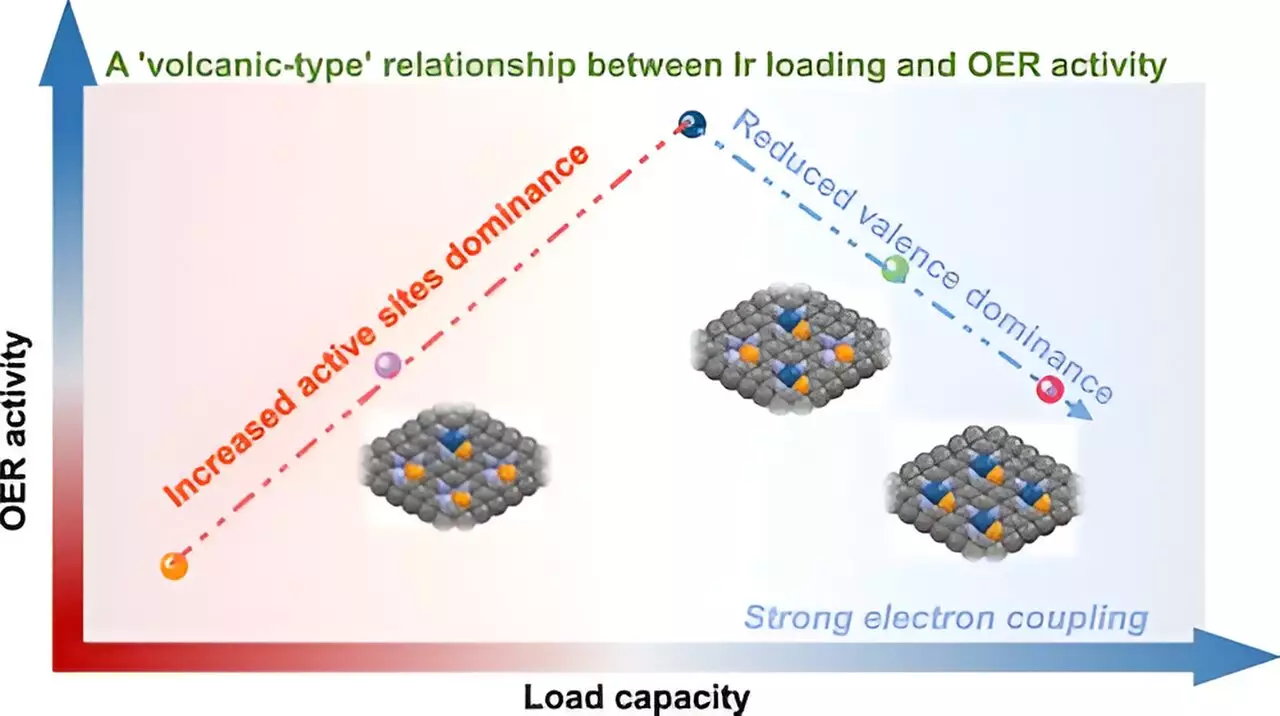
Single-atom catalysts (SACs) present a revolutionary approach within catalysis by offering a high degree of efficiency derived from their unique properties. However, one of the major technical challenges has been maintaining atomic-level dispersion while also increasing metal loadings. The USTC team’s exploration into the “volcano-type” relationship regarding the efficacy of these catalysts provides a nuanced understanding of how to navigate these challenges. Traditional views often led researchers to assume a straightforward positive correlation between metal loading and catalytic activity. Yet, this emerging paradigm challenges that notion.
The Volcano Effect: A Paradigm Shift
The so-called “volcano-type” relationship offers a compelling narrative: as metal loading increases, initially, there is a corresponding rise in the catalyst’s activity due to the greater availability of active sites. However, this relationship is not linear. Beyond a certain threshold—identified in the USTC study—it becomes increasingly counterproductive; the interactions among the adjacent metal atoms change significantly, detracting from performance as the catalyst nears its limits.
Through advanced techniques involving synchrotron radiation X-ray absorption spectroscopy (XAS) and X-ray photoelectron spectroscopy (XPS), the researchers illuminated the fundamental interactions at play. The critical innovation lies in the Ir-P coordination structure that effectively stabilizes iridium atoms, preventing harmful aggregation that typically undermines the performance of catalysts at higher loadings. This achievement signals a potential game-changer in the design of more efficient catalytic systems, particularly in the realm of oxygen evolution reactions (OER), which are vital for energy applications like water splitting.
The Future of Catalyst Design
What makes this research particularly exciting is its forward-looking perspective. By unraveling the complexities of single-atom catalysts and understanding their interactions at a microscopic level, the team has laid the groundwork for future enhancements in catalyst design. As the demand for efficient catalysts grows—driven by the needs of renewable energy and environmental remediation—these findings can guide the development of new materials that harness the strengths of single-atom catalysts while avoiding pitfalls associated with excess metal loading.
It’s imperative that the scientific community embraces this “volcano-type” relationship as it drives further exploration into optimal catalyst design. This research not only contributes to theoretical frameworks but also provides actionable insights for engineers and developers working on practical applications. With the right materials and strategies in hand, the potential for energy solutions that are both effective and economically viable is closer than ever. The dialogue about single-atom catalysis is transforming, and the stakes couldn’t be higher for our energy future.

Leave a Reply