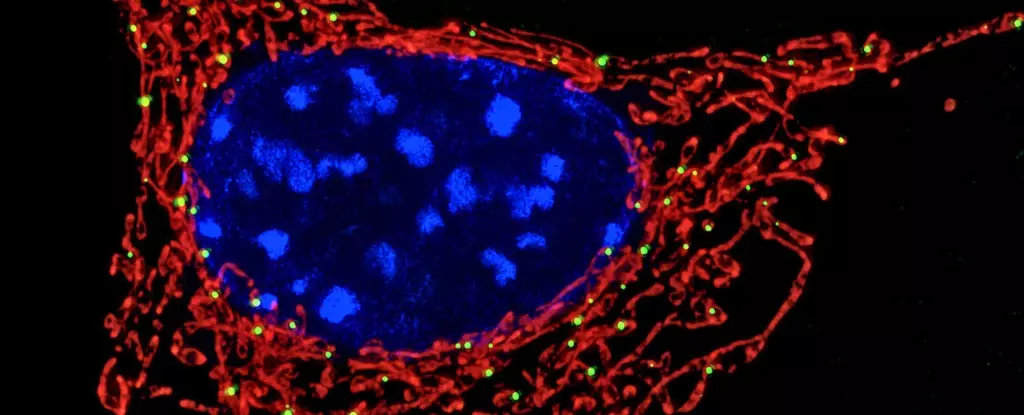
Mitochondria have long been simplified as the cell’s “powerhouses,” primarily responsible for generating the energy required for life. Yet, this narrow perspective ignores an expanding body of evidence positioning mitochondria at the core of immune regulation. Recent research, including pioneering studies on neutrophils—the most abundant white blood cells—shows mitochondria act as sophisticated sensors that detect microbial invaders and orchestrate immune defenses. This emerging view challenges traditional immunology by integrating metabolism and immune function into a unified framework, painting mitochondria as crucial immune sentinels rather than mere bioenergetic factories.
The Discovery of Metabolic Signaling in Immune Defense
The immune system’s innate cells must rapidly recognize threats and respond appropriately. Neutrophils, as first responders, employ diverse tactics to neutralize pathogens. One such tactic is the deployment of neutrophil extracellular traps (NETs)—sticky DNA structures imbued with antimicrobial proteins that ensnare bacteria. Until recently, NET formation was loosely attributed to cellular stress. However, new findings reveal a far more nuanced trigger: mitochondria detect lactate, a metabolic byproduct excreted by bacteria, within phagosomes (intracellular compartments). This metabolic cue acts as a molecular beacon indicating live bacterial presence and insufficient intracellular killing, prompting neutrophils to release NETs and externalize the bacteria for elimination by other immune cells.
This discovery highlights a startling level of intracellular communication: mitochondria, residing outside the phagosome, somehow perceive biochemical signals within these enclosed compartments. Such intricate cross-talk underscores the complexity of cellular immune defenses, where metabolic signals direct precise immune responses rather than blunt reactions to generic damage.
Implications for Autoimmune Conditions and Infection Susceptibility
A particularly striking aspect of this mitochondrial sensing mechanism emerges in the context of autoimmune diseases such as systemic lupus erythematosus (SLE). Paradoxically, despite persistent immune activation characteristic of lupus, patients often experience recurrent infections. Investigations reveal that the mitochondria in lupus neutrophils fail to recognize bacterial lactate effectively, resulting in impaired NET formation. This mitochondrial dysfunction bridges two seemingly contradictory phenomena: immune system overactivity and immunodeficiency.
This insight not only explains clinical observations that had long puzzled researchers but also reframes mitochondria as pivotal modulators of immune balance—capable of both ramping up defensive responses when functioning properly and contributing to immune dysregulation when impaired.
Redefining Immunometabolism: Mitochondria at the Crossroads
The field of immunometabolism has gained traction by acknowledging that metabolism is far from a passive background process; it is a dynamic driver shaping immune cell behavior. The mitochondrial capacity to sense metabolite signals such as bacterial lactate integrates energy production with immune signaling in a feedback loop that dictates immune outcomes. This discovery redefines mitochondria as metabolic orchestrators, central to deciding when and how immune defenses should be activated.
For example, manipulating mitochondrial sensing pathways could revolutionize therapeutic approaches. Enhancing mitochondrial detection of pathogen metabolites could bolster immunity in vulnerable populations prone to infections, such as those with lupus or other immunocompromising conditions. Conversely, moderating excessive NET formation could alleviate tissue damage resulting from overzealous immune responses observed in diseases like severe COVID-19 or chronic autoimmune disorders.
Future Horizons: Expanding the Landscape of Metabolic Immune Sensing
While lactate detection by mitochondria presents a groundbreaking advance, it opens the door to numerous questions. Do other immune cells utilize comparable mechanisms to monitor microbial metabolism? Are additional bacterial byproducts employed as immunological messengers? Deciphering these pathways promises to refine our ability to fine-tune immune responses, minimizing collateral damage while maximizing pathogen clearance.
In unraveling mitochondria’s multifaceted roles—from energy provision to immune oversight—science is moving towards a holistic understanding of cellular systems. Mitochondria serve not only as energy sources but as vigilant metabolic watchdogs, continuously interpreting subtle biochemical signals that tell the immune system when to act. Their strategic placement at the intersection of metabolism and immunity positions them uniquely to shape health outcomes and disease progression.
A Critical Perspective: Recognizing the Complexity and Limitations
Despite these exciting advances, we must remain cautious not to oversimplify mitochondria’s functions. While the discovery of lactate sensing in neutrophils is compelling, immune regulation involves layers of overlapping signals and pathways. The intracellular environment is a maze of interactions, and isolating mitochondrial roles risks neglecting the contributions of other organelles, signaling molecules, and systemic factors.
Furthermore, translating these findings into clinical interventions demands rigorous testing. Therapies targeting mitochondrial sensing must balance efficacy with safety, avoiding unintended consequences such as exacerbated inflammation or compromised immunity. The interconnectedness of immunometabolism means that altering one component might ripple unpredictably through the system.
In embracing mitochondria as linchpins of immune defense, the scientific community embarks on a nuanced journey—one that will require integrating cellular biology, metabolism, and clinical insights to harness these tiny organelles’ full therapeutic potential.

Leave a Reply