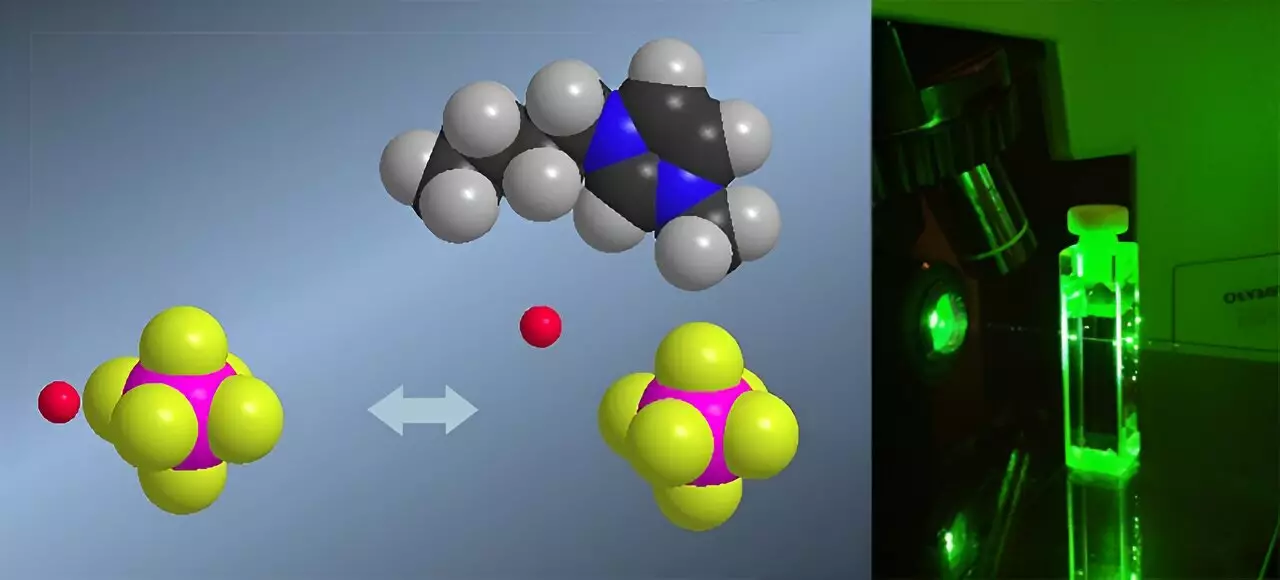
In the realm of modern chemistry, ionic liquids have long presented a perplexing challenge: how to accurately measure their acidity. Unlike conventional aqueous solutions, these organic salts remain liquid at room temperature and exhibit extraordinary acidity—sometimes up to 100 million times more potent than water-based acids. Traditional pH measurements falter here, as standard methods depend on the solvent’s transparency and their compatibility with colored indicators, both of which ionic liquids do not reliably offer. Highlighting a significant breakthrough, researchers at the University of Liège have pioneered the first application of Raman spectroscopy to determine the Hammett acidity function in these complex media. This is not just a technical feat; it critically enhances our capacity to understand, predict, and manipulate reactions within ionic liquids—an advancement with profound scientific and industrial implications.
Why Raman Spectroscopy Changes the Game
Unlike the conventional UV-visible spectroscopy techniques, Raman spectroscopy provides a versatile, error-resistant way to measure acidity in ionic liquids. Its non-reliance on optical transparency or colored indicators means that the often opaque, colorless, or interactively complex environments of ionic liquids are no barrier. This innovative approach enables researchers like Aurélie Rensonnet to obtain more precise and reliable data on acidity levels, an essential step forward for anyone interested in the catalytic or electrochemical applications of these solvents. By confirming the ability to accurately measure Hammett acidity functions in ionic liquids, the study significantly expands the horizon for future research into their chemical behaviors and potential uses.
Implications for Industry and Scientific Understanding
This breakthrough opens the door to a better grasp of acidity-sensitive reactions involving ionic liquids. Previously, the inability to measure how protons transfer and behave in these environments limited our understanding of their chemistry. Now, scientists can experimentally estimate the energy needed for proton transfer from water to ionic liquids, complementing computational models in a synergistic dance that deepens our understanding of these unique solvents. Such insights are invaluable for industries seeking to optimize processes like acid catalysis, battery electrolyte development, and biomass depolymerization—technologies that hinge on precise acidity control. Knowing how super-acidic ionic liquids behave could revolutionize how we design sustainable energy solutions and efficient chemical conversions.
Transformative Potential and Future Directions
This pioneering research not only pushes the boundaries of laboratory chemistry but also heralds a new era where ionic liquids can be tailored with unparalleled precision. The ability to determine acidity on-site and in real time means safer, faster, and more efficient process optimization in various fields—from pharmaceuticals to renewable energy. Moreover, the collaboration between experimental measurements and computational chemistry will foster a deeper understanding of proton transfer dynamics, enabling smarter, more predictive models. As scientific curiosity continues to push beyond old limits, the work from Liège signals a promising frontier: one where the enigmatic nature of ionic liquids becomes increasingly transparent, giving scientists and industry leaders a powerful tool to harness their full potential.

Leave a Reply