Carboxylic acids occupy a pivotal spot in the realm of organic chemistry, being integral to various essential compounds, notably pharmaceuticals like aspirin and ibuprofen. Their unique chemical properties and versatility allow them to participate in a myriad of chemical reactions, making them valuable to researchers and industries alike. The need for enhancing these properties has led chemists to explore ways to modify their structure, one exciting avenue of which involves the incorporation of fluorine into the molecular framework. Fluorinated compounds exhibit unique attributes, such as increased lipophilicity and altered biological activity, which can drastically improve the effectiveness of medicinal agents.
The Complexity of Traditional Fluorination Techniques
Historically, the introduction of fluorine into the structure of carboxylic acids has been a challenging and intricate process, often necessitating elaborate multi-step synthetic routes. These methods not only demand significant time and resources but also carry the risk of producing unwanted by-products, complicating the purification process. This complexity serves as a barrier to the efficient development of new fluorinated drugs, underscoring the necessity for innovative strategies that simplify and streamline synthesis.
A Breakthrough Methodology
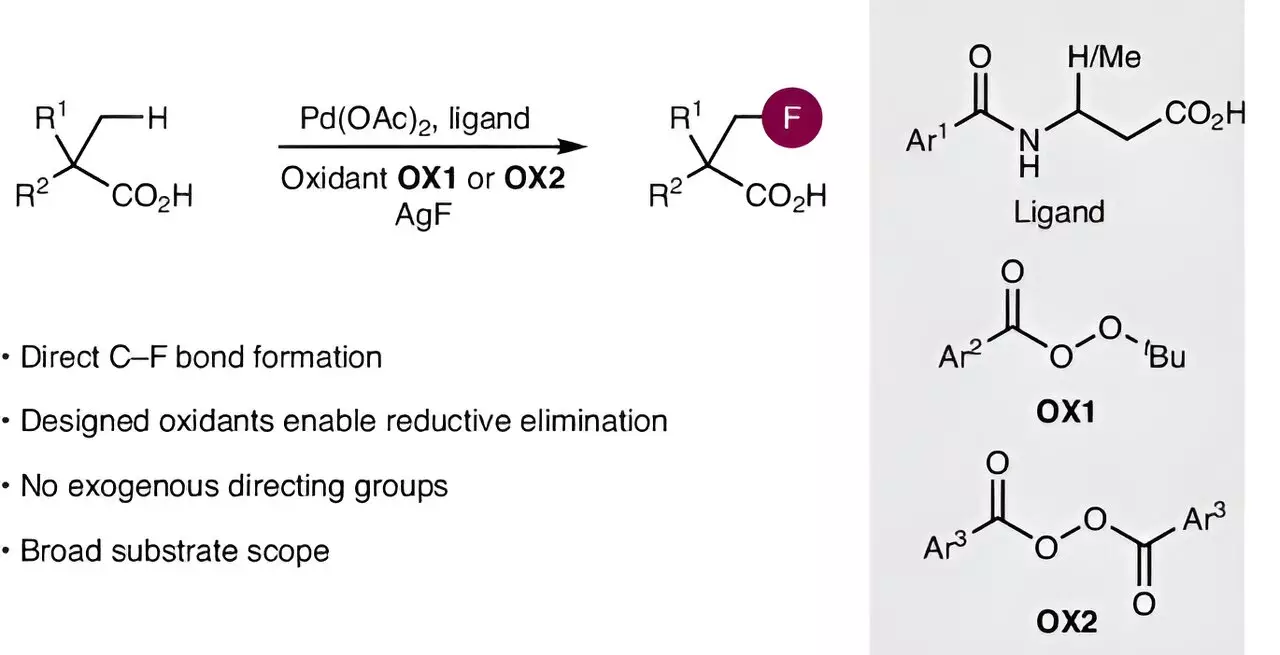
Researchers at the Otto Diels Institute of Organic Chemistry at Kiel University, led by Professor Manuel van Gemmeren, have recently made significant strides in addressing these challenges. Their newly published work in *Nature Synthesis* introduces a revolutionary method that allows for the direct fluorination of aliphatic carboxylic acids, thus obviating the need for cumbersome traditional techniques. This new approach hinges on activating the notoriously inert carbon-hydrogen (C–H) bonds, enabling chemists to facilitate the critical carbon-fluorine (C–F) bond formation.
The activation of C–H bonds is a complex undertaking typically requiring advanced catalysts. Van Gemmeren’s team has capitalized on recent breakthroughs in catalyst development to construct more efficient palladium-based systems. Their approach not only streamlines the introduction of fluorine but also sets the stage for a more generalizable technique that could positively impact the synthesis of various other organic compounds.
Innovative Solutions for Transformation
Moreover, the researchers faced the daunting task of establishing a suitable means of forming a robust C–F bond in the carboxylic acid framework. Given that conventional fluorination strategies were ineffective for their objectives, they engineered a tailored oxidizing agent designed to synchronize with the catalyst, promoting selective reactions. This innovative collaboration between catalyst design and reagent functionality represents a paradigm shift in fluorination chemistry, suggesting that such integrated strategies could have widespread applicability across multiple synthetic disciplines.
Implications for Pharmaceutical Development
The implications of this research are profound, especially considering the crucial role that both carboxylic acids and fluorinated compounds play in medicinal chemistry. By simplifying the fluorination process, this methodology can expedite the development of new drug candidates, potentially leading to more effective therapies with optimized properties. As the pharmaceutical industry continues to seek novel compounds that enhance therapeutic efficacy, this groundbreaking technique promises to play a significant role in future research endeavors, driving innovation in drug design and development.

Leave a Reply