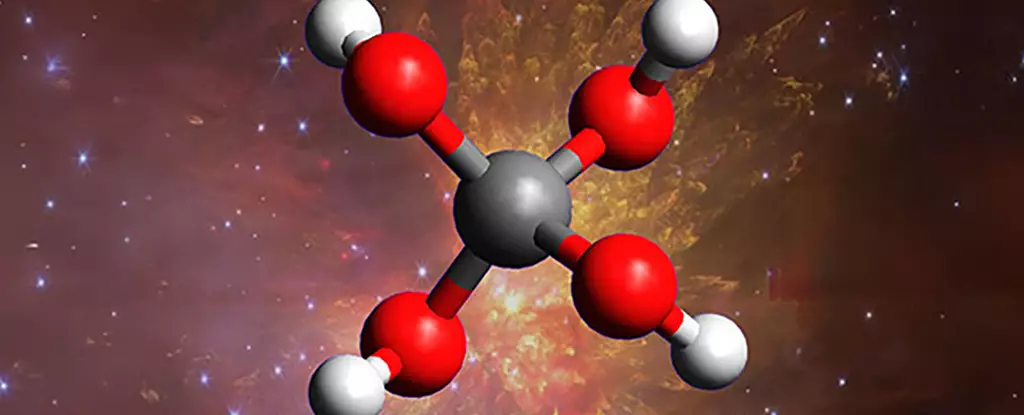
In a remarkable scientific triumph, researchers have successfully synthesized a long-theorized yet elusive molecule—methanetetrol (C(OH)₄)—within laboratory conditions that mimic the extreme environments of interstellar space. This discovery not only confirms a century-old prediction but also radically redefines our understanding of the complex chemistry taking place in the cold, dark reaches of the universe. The ability to create such an unstable super alcohol under controlled conditions offers profound insights into the potential chemical diversity hidden within interstellar clouds, expanding the horizons of astrobiology and molecular astrophysics.
Much of what we know about space chemistry has been built on observations of relatively simple molecules, such as water, methane, and formaldehyde. However, the identification of methanetetrol—a molecule comprising four hydroxyl groups attached to a single carbon atom—upends this limited perspective. The molecule’s extraordinary instability explains why it has evaded detection in the natural cosmos; it disintegrates rapidly when exposed to light, making it an ephemeral entity that challenges current detection techniques. Yet, its synthesis in a laboratory setting demonstrates that the formation of such molecules is not only possible but perhaps more common than previously thought, especially within the cold, radiation-rich environments of interstellar ice clouds.
The experimental approach undertaken by the international team was groundbreaking. By creating artificial space ice—freezing carbon dioxide and water at cryogenic temperatures in vacuum chambers—they recreated the conditions found in the dense molecular clouds that serve as stellar nurseries. Bombarding this ice with high-energy radiation, mimicking the cosmic rays that pervade space, initiated complex chemical reactions. Under these simulated cosmic conditions, methanetetrol emerged momentarily before disintegrating, providing concrete proof of its potential existence beyond Earth’s laboratory. This process underscores how energetic radiation acts as a cosmic catalyst, facilitating the formation of complex molecules in environments that are seemingly inhospitable.
This breakthrough raises critical questions about the richness of space chemistry. If molecules as complex and unstable as methanetetrol can form naturally, the diversity of chemical compounds in the universe may be vastly underestimated. The implications extend beyond mere molecular cataloging; they may influence our understanding of the pathways leading to prebiotic chemicals—the precursors of life. Could these fleeting molecules serve as intermediates in the synthesis of biologically relevant compounds? If so, the origins of life might be more intimately connected with the chemistry of the cosmos than previously assumed.
Moreover, the discovery acts as a catalyst for a paradigm shift in the search for extraterrestrial life. The universe could harbor complex, yet transient, molecules that act as stepping stones toward biologically significant substances. This hints at a universe teeming with capabilities that have been largely hidden from our detection methods due to the molecules’ instability. The challenge now lies in developing technologies capable of capturing and analyzing these transient chemical signatures directly in space. The hope is that, as telescope sensitivities improve and spectroscopic methods advance, we may eventually detect such fleeting molecules in their natural habitats—deep within dense clouds or around dying stars.
Despite the excitement, significant hurdles remain. Methanetetrol’s delicate nature means it cannot survive in Earth’s warmer, light-friendly conditions; thus, any direct observation in space remains technologically demanding. The molecule’s rapid dissociation under illumination—caused by photosensitive destruction—complicates its detection. Nevertheless, this research serves as an essential stepping stone, informing astronomers where to focus their efforts and what signatures may indicate the presence of similar complex molecules. The pioneering work illustrates that our previous limitations, primarily due to the molecules’ instability, are now being systematically challenged and overcome.
In the broader context, this breakthrough underscores that the universe’s chemistry is far more intricate and dynamic than our current models suggest. We have only scratched the surface of the cosmic chemical inventory—estimating that less than 1% of space’s chemical diversity has been uncovered. Each new discovery nudges us closer to understanding the intricate dance of molecules in space, revealing how the building blocks for life might be synthesized in environments once considered too extreme or too simple. The synthesis of methanetetrol fuels optimism that many more complex, ‘impossible’ molecules await discovery—hidden in cold dust clouds or formed transiently in the aftermath of cosmic explosions.
The ongoing exploration of space’s chemical landscape is not merely a pursuit of curiosity; it could redefine our understanding of life’s origins and the potential for life beyond Earth. With each molecular breakthrough, we get closer to unraveling the cosmic recipe for life, revealing a universe more chemically vibrant and fundamentally creative than we ever imagined. Methanetetrol stands as a symbol of scientific ingenuity—proof that some of the universe’s most elusive secrets are within reach, hidden within the cosmic tapestry of molecules that form in the shadows of stars and the depths of interstellar ice.

Leave a Reply