Mechanophores—molecules that alter their chemical or physical properties under mechanical stress—have emerged as powerful tools in developing responsive materials, advancing organic synthesis, and innovating drug delivery systems. Among recent breakthroughs is the discovery of NEO, a mechanophore by University of Illinois Urbana-Champaign chemists, notably Prof. Jeffrey Moore and graduate student Yunyan Sun. NEO’s remarkable ability to release controlled doses of carbon monoxide upon mechanical triggering offers tantalizing potential, especially for targeted therapeutics inside the human body. Yet, the scientific community faces a major obstacle in reliably predicting how these molecules, particularly in the breaking of carbon-carbon (C–C) bonds—a fundamental process in mechanochemistry—respond to force. The inherent complexity arises from the vectoral nature of applied forces and the unpredictable pathways of molecular rupture, often demanding extensive experimental work and computational resources to understand.
A Breakthrough: The Emergence of TMBA
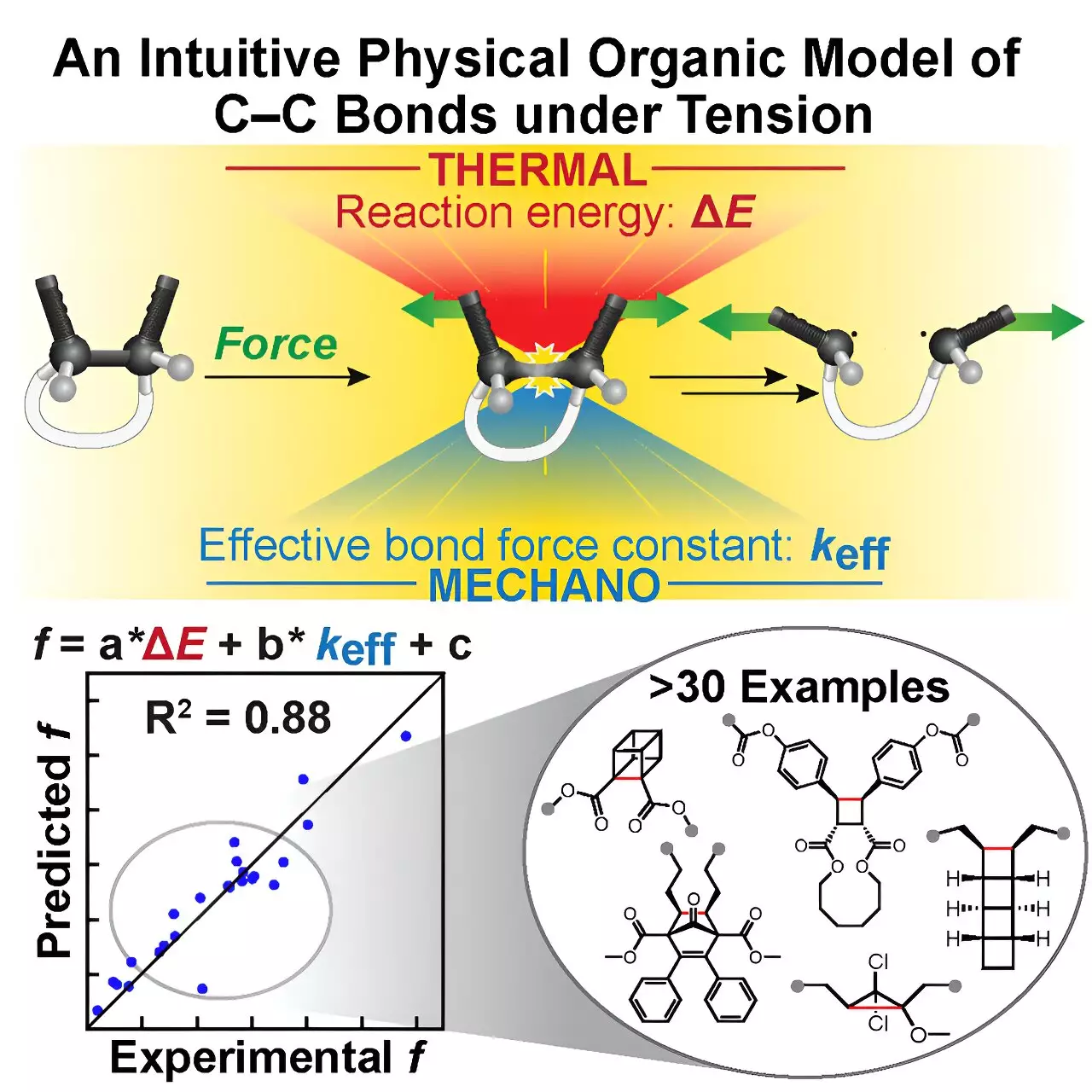
In a significant stride forward, Moore and Sun, collaborating with researchers from MIT and Duke University, introduced an elegant, intuitive computational tool known as the Tension Model of Bond Activation (TMBA). Based on an age-old, simple chemical principle—the Morse Potential describing interatomic interactions—this mnemonic framework distills the intricacies of C–C bond breaking into easily calculated parameters: the effective force constant and the reaction energy. By conceptualizing these using a geometric “restoring force triangle,” TMBA translates elaborate mechanochemical behaviors into manageable, quantifiable insights. This model doesn’t just simplify the math; it bridges the gap between high-level theoretical outputs and tangible molecular design strategies. The ability to intuitively grasp mechanophore reactivity without the usual heavy computational burden is poised to reshape how chemists approach mechanochemistry.
The Importance of Intuition in Mechanochemical Design
Yunyan Sun aptly underscores the value of TMBA as a discovery tool that complements, rather than replaces, sophisticated simulations. Traditional computational chemistry, while precise, often remains inaccessible without specialized knowledge and can obscure practical understanding amid complex data. TMBA’s strength lies in its teachability and clarity, rendering it attractive for chemists at all levels who need to strategize without blindly relying on high-end computations. By providing quick, understandable predictions, the model serves as a conceptual compass, guiding experimentation and hypothesis formulation on mechanophore behavior. This approach echoes the foundational role of simplified models in chemistry, like Lewis structures or Marcus theory, which distill complex phenomena into principles that are both intuitive and empirically useful.
From Unexpected Observations to Universal Principles
The conception of TMBA came from serendipity—studying anomalous reactivity trends in a series of NEO mechanophores sparked curiosity and deeper inquiry. Sun and her colleagues observed that disparate mechanophores shared underlying commonalities governed by specific molecular properties. Investigating these patterns propelled the team beyond isolated cases toward a generalized framework that resonates with broader mechanochemical behavior. Interdisciplinary collaboration was central here; ideas originated from Duke’s Prof. Steve Craig’s lab, and the collective effort culminated in a linear predictive model validated across more than 30 known mechanophores. This partnership exemplifies how cross-institutional synergy fuels innovation, combining unique expertise and methodologies to solve complex challenges in material science.
Transformative Implications for Science and Education
The significance of TMBA extends beyond basic research—it has the potential to accelerate the discovery of new mechanophores producing novel mechanically responsive materials. Researchers envision integrating this tool into large-scale computational screenings, effectively filtering promising candidates by predicted reactivity before experimental validation, thus saving time and resources. Furthermore, the model’s simplicity facilitates its adoption in educational settings, potentially becoming a staple in undergraduate curricula. This democratization of mechanochemical insight disrupts the prior dogma that C–C bonds are virtually unbreakable—a belief that TMBA robustly challenges. By shifting perceptions and lowering barriers to entry, the field moves toward a productive mechanochemistry era, where mechanical forces can be precisely tailored to achieve desired molecular transformations.
Interdisciplinary Collaboration: The Engine Driving Innovation
The TMBA project was conceived and realized only through the collaborative interplay of chemists and engineers from different research institutions, illustrating the profound impact of teamwork in advancing mechanochemistry. The MONET Center (Center for the Chemistry of Molecularly Optimized Networks) embodies this philosophy by uniting specialists whose complementary skills and perspectives enriched the research. Duke’s Stephen L. Craig and Tatiana Kouznetsova contributed critical single-molecule experimental data, seamlessly merging empirical and theoretical approaches. This collective intelligence was essential in refining the model, corroborating predictions, and extending TMBA’s applicability. The project underscores a broader lesson in contemporary science: multifaceted challenges demand diverse minds and shared expertise.
A New Paradigm for Understanding and Designing Mechanochemical Reactions
The research spearheaded by Moore, Sun, and their collaborators signals a paradigm shift. By translating the complex world of mechanochemical bond breaking into a simple, teachable framework, TMBA equips researchers to better predict, interpret, and ultimately harness molecular responses to mechanical stress. The implications are profound—not just for mechanophores but for mechanochemistry at large, enabling more rational design of materials whose function originates from mechanical stimuli. As mechanochemistry evolves from a niche curiosity into a mainstream field, tools like TMBA democratize knowledge, empower innovation, and challenge existing chemical dogma with clarity and elegant simplicity. This is science at its best: breaking down complexity to unlock new possibilities.

Leave a Reply