In recent strides within organic chemistry, researchers from Tokyo Tech have unveiled a groundbreaking synthesis strategy that could revolutionize the way we create complex molecular frameworks. This novel approach utilizes inexpensive quinolines as the primary feedstock, which has potential implications for drug discovery and custom pharmaceuticals. By harnessing the properties of a light-sensitive borate intermediate, the scientists have devised a method to transform quinoline derivatives into a myriad of fused 2D/3D frameworks. The implications of this discovery, detailed in *Angewandte Chemie International Edition*, are monumental, hinting at an era where creating highly customizable drug candidates becomes more feasible than ever.
Quinolines: The Unsung Heroes of Molecular Synthesis
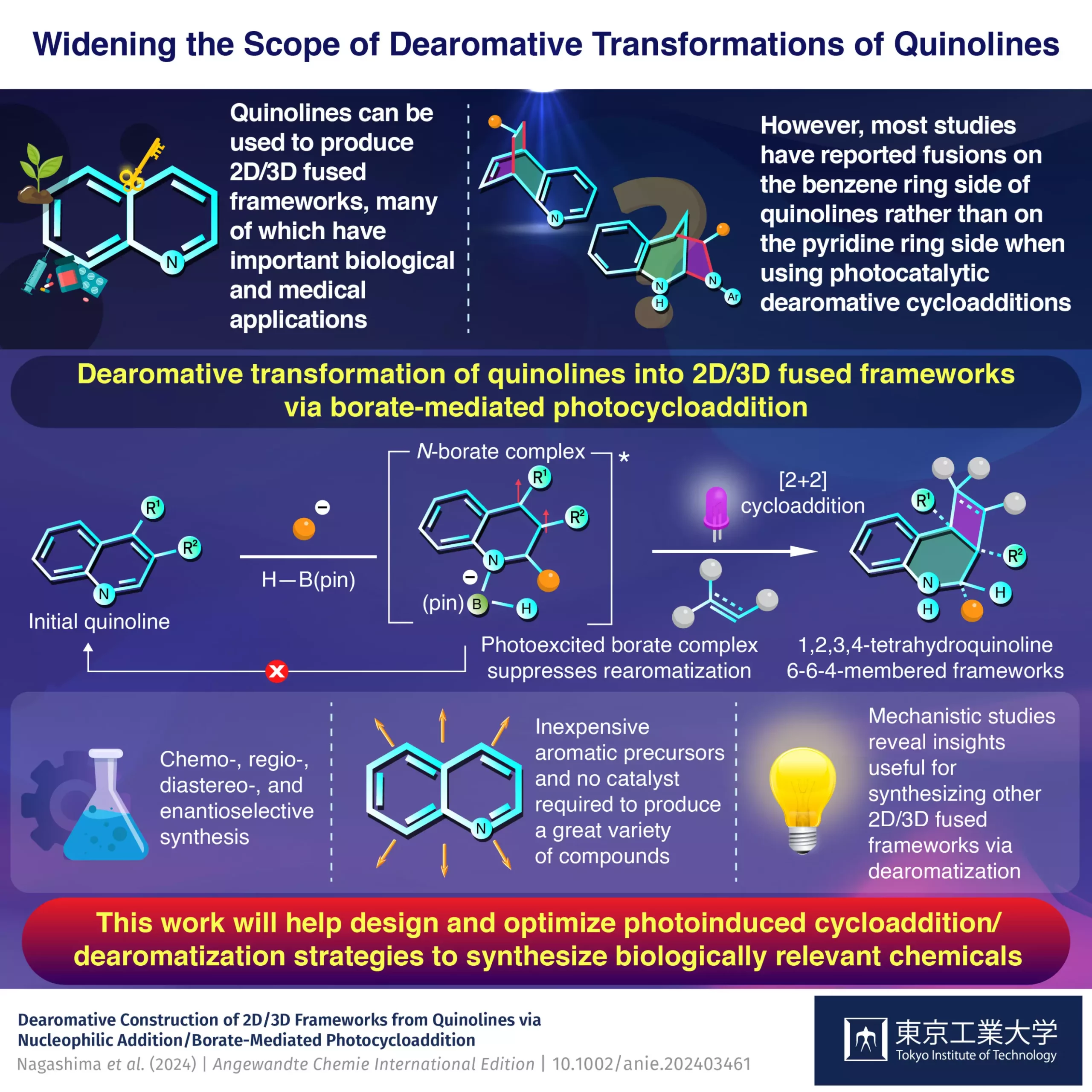
Quinolines have long been recognized as valuable compounds in the synthetic chemist’s arsenal, primarily due to their unique electronic structure. With an aromatic benzene ring fused to an electron-deficient pyridine ring, quinolines possess properties that allow chemists to independently modify each component by tweaking reaction conditions. This aspect of their chemistry becomes particularly intriguing when considering the potential applications in medicine, as these modifications can lead to highly effective therapeutic agents. However, the challenge has traditionally been in accessing the pyridine side of quinolines for synthetic purposes, particularly through methods such as dearomative photocycloadditions, which have predominantly targeted the benzene side.
Redefining Reaction Strategies
The Tokyo Tech team, led by Assistant Professor Yuki Nagashima, seized this challenge head-on and has made significant advancements. Their research introduces a groundbreaking method that primarily focuses on the pyridine side of quinoline, effectively unlocking a previously underutilized aspect of the molecule’s reactivity. At the heart of their approach is pinacolborane, a boron-containing compound that effectively enables the desired photocycloaddition reactions. By doing so, they not only achieved high yields but also showcased the versatility of different quinoline derivatives, easily expanding the potential library of 2D/3D frameworks that can be synthesized.
The researchers’ method stands out because it reduces the time and complexity typically associated with traditional synthesis routes. By eschewing the need for catalysts and enabling the use of diverse starting materials, this approach promises to be both more efficient and cost-effective, making it particularly appealing for larger-scale pharmaceutical applications.
A Novel Mechanistic Insight
A significant factor in the success of this new synthetic strategy lies in the scientists’ profound understanding of the underlying mechanisms. Through a series of theoretical analyses and experiments, they established that the reaction begins with quinoline engaging with an organolithium compound, subsequently forming a borate complex with H–B(pin). This intermediate complex is crucial, as it simultaneously promotes the cycloaddition while minimizing the tendency for rearomatization—a common pitfall in conventional reactions. Assistant Professor Nagashima articulates the importance of this discovery, underscoring how the photoexcited borate complex accelerates cycloaddition and leads to higher yields of desired products.
Such mechanistic insights not only enhance the understanding of quinoline chemistry, but they also open doors to future research that could further exploit organoboron compounds. The realization that these transformations are the first of their kind in boron-based photocycloadditions marks a significant milestone, shedding light on how we might manipulate complex organic structures with greater precision.
The Road Ahead: Implications for Drug Development
As the scientific community turns its gaze toward the therapeutic applications of these newly synthesized compounds, the potential implications are exhilarating. The customization of drug candidates through this innovative synthesis method could lead to the development of therapies tailored to individual biological pathways, enhancing effectiveness while reducing side effects. The strategic advantages provided by the Tokyo Tech research team’s methodology could well spur a new wave of explorations into multi-ringed aromatic hydrocarbons.
Overall, this synthesis strategy signals a pivotal shift in how chemists can leverage the unique properties of quinolines. By aligning cutting-edge chemistry with economic considerations, researchers are not merely expanding our foundational knowledge—they are creating new pathways towards tangible medical advancements. As more scientists harness these insights, the future of organic synthesis looks promising, propelling us closer to transformative solutions in medicine.

Leave a Reply