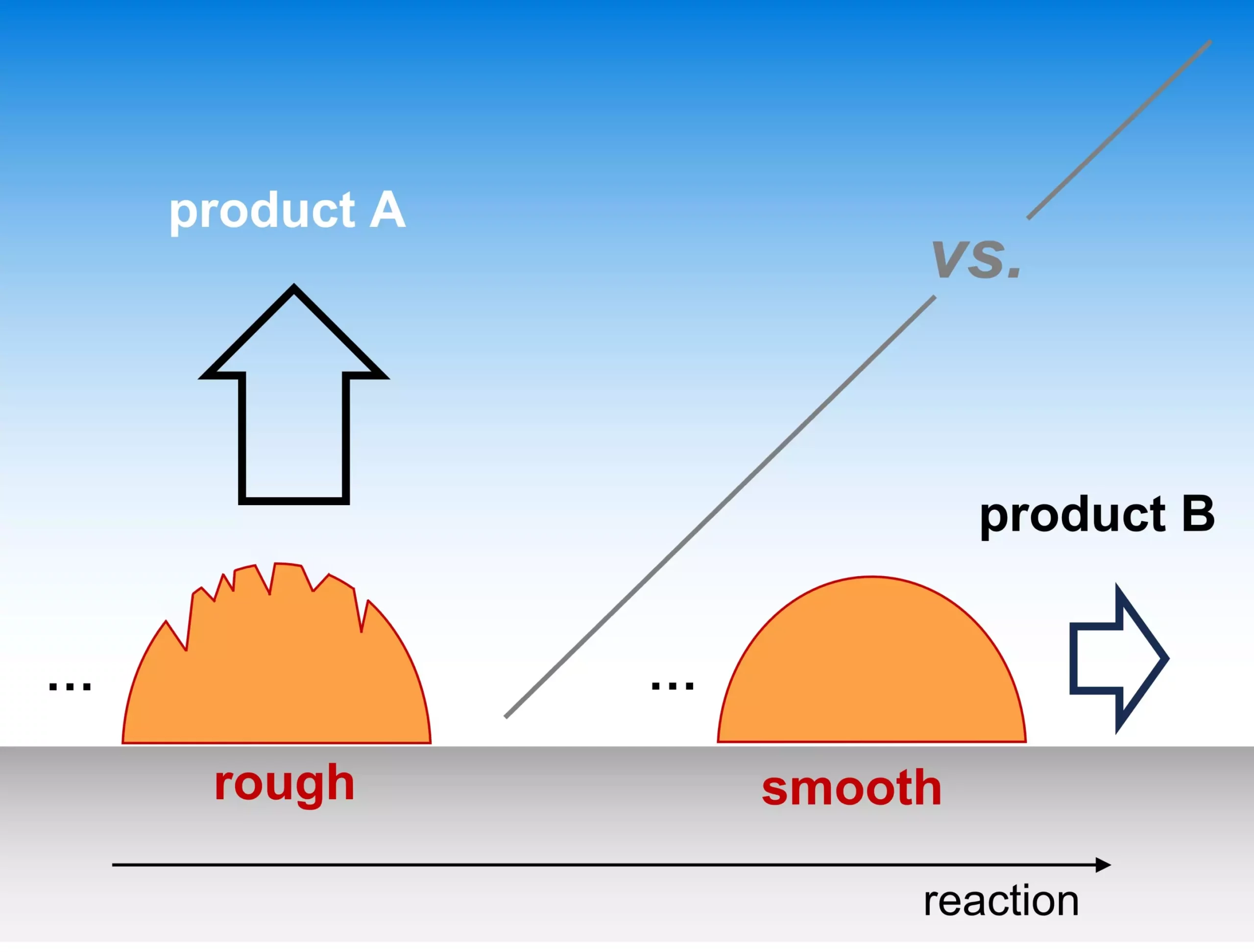
The Fritz Haber Institute’s Theory Department has opened a significant dialogue regarding the role of catalyst morphology in electrocatalytic reactions. Their recent findings, prominently featured in *Nature Catalysis*, demonstrate how the surface characteristics of a catalyst—specifically its “roughness”—play a crucial role in determining which products are generated during these complex chemical processes. This innovative perspective shifts the emphasis from merely focusing on atomic-level active sites to a more nuanced understanding of how varying surface textures can influence reaction outcomes.
Significance for Sustainable Technologies
Catalysis is indispensable to modern chemical industries; its applications extend from the production of plastics and pharmaceuticals to the creation of fertilizers. Particularly, heterogeneous electrocatalysis stands at the forefront of sustainable energy efforts, enabling the green production of fuels and chemicals driven by renewable energy sources. The implications of this research extend far beyond academic interest; they hold the potential to transform how we conceive of and implement sustainable practices, as these reactions can occur under mild conditions of temperature and pressure, primarily relying on charge transfer at the solid-liquid interface.
Challenges of Catalyst Selectivity
Yet, despite its importance, the intricacies of catalyst selectivity remain inadequately understood, particularly within electrocatalysis. Selectivity, which dictates the efficiency and effectiveness of various reactions, has long puzzled scientists. The work of the Fritz Haber Institute proposes a new avenue for exploration, focusing on the microscopic mechanisms at play. For instance, they elucidate how certain reaction intermediates can escape the catalyst surface, affecting the detection of partially-converted products. By delving deeper into these processes, we may unravel the complexities underlying selectivity and begin to develop more efficient systems.
A Multi-scale Approach to Kinetic Modeling
A standout aspect of the research is the introduction of a multi-scale kinetic model that quantitatively assesses how selectivity varies with different factors, particularly the transport rates of reaction species through the electrolyte and the density of catalytically active sites. This model showcases its utility by reproducing trends observed in experimental literature, thus validating its application and enhancing our understanding of when and why certain reactions prevail over others.
Transformative Insights for Future Research
The emphasis on catalyst roughness as a defining feature of morphology paves the way for further investigations into the optimization of electrocatalytic processes. This revelation not only enriches our fundamental grasp of reaction mechanisms but also inspires innovative strategies for advancing catalyst design. By synergizing theory with practical applications, these insights may contribute significantly to achieving greater efficiency and reliability in sustainable energy technologies.
The innovative work from the Fritz Haber Institute sheds light on a critical yet often-overlooked aspect of catalysis, offering a fresh perspective that could revolutionize our approach to sustainable chemical processes. The emphasis on catalyst morphology and its influence on selectivity represents a compelling area of research that promises to yield transformative results in the pursuit of clean energy solutions.

Leave a Reply